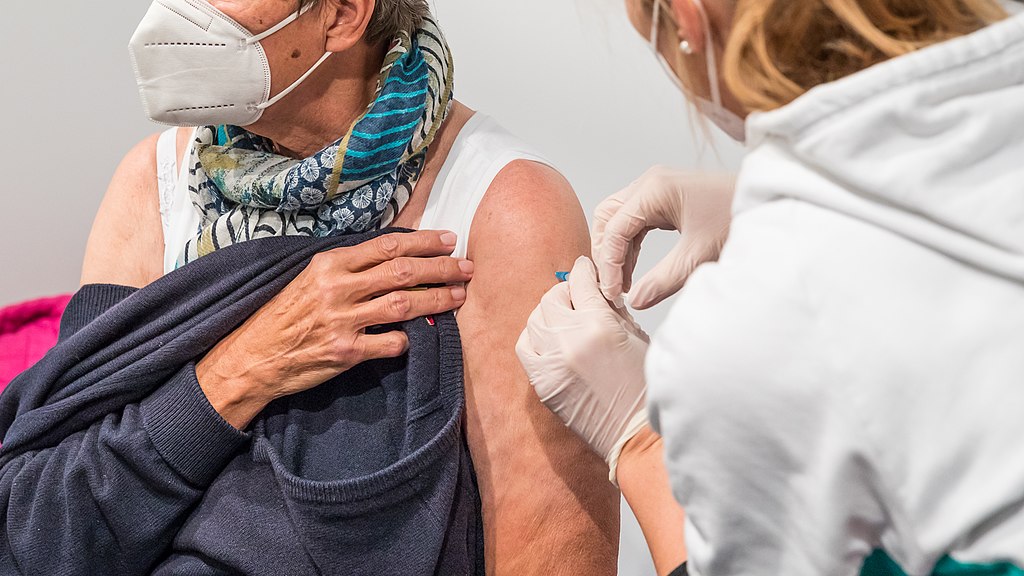
The Food and Drug Administration announced it is simplifying its recommended COVID-19 vaccination schedule, saying there is no need for multiple vaccinations and boosters.
The FDA will now recommend a single dose of the bivalent vaccine specific to two strains of the omicron variant.
Its decision reflects a shift away from the two-dose schedule that is currently the standard across the country. During the peak of the pandemic, federal agencies recommended (and in some cases, mandated) a two-dose vaccination followed by multiple boosters.
“At this stage of the pandemic, data support simplifying the use of the authorized mRNA bivalent COVID-19 vaccines and the agency believes that this approach will help encourage future vaccination,” Peter Marks said in a statement.
Marks, the director of the FDA’s Center for Biologics Evaluation and Research, said most of the US population now has antibodies against the virus from either prior infection or previous vaccinations.
The FDA will continue to recommend two doses for some groups, such as people over the age of 65 and immunocompromised people, but it advises those doses are given two to four months apart.
During the height of the pandemic, doses were given just three weeks apart with boosters for new strains within months.
Serious Issues Swept Under the Rug
Heart problems, in some case fatal, became a notable side effect of the frequent vaccination schedule during the pandemic.
The CDC reports they occurred most frequently in adolescent and young adult males, but were also documented in individuals of both genders who were otherwise perfectly healthy and, in some cases, athletes in peak physical condition.
“Evidence from multiple monitoring systems in the United States and around the globe support a causal association between mRNA COVID-19 vaccines (i.e., Moderna or Pfizer-BioNTech) and myocarditis and pericarditis,” the CDC says.
“Cases of myocarditis and pericarditis have occurred most frequently in adolescent and young adult males within 7 days after receiving the second dose of an mRNA COVID-19 vaccine; however, cases have also been observed after dose 1 and booster doses.”